The key difference between Bohr and Schrodinger model is that in the Bohr model, electrons are particles that occupy only certain orbits of fixed energy around the nucleus, whereas, in the Schrodinger model, electrons behave as standing waves having a greater probability of being in some areas of space compared to others.īelow is a summary of the difference between Bohr and Schrodinger model in tabular form for side by side comparison.īoth the Bohr model and Schrodinger model describe the chemical structure of atoms and the location of electrons at a given time. What is the Difference Between Bohr and Schrodinger Model?īohr model and Schrodinger model are like the basics of general chemistry. He combined the equations for the behavior of waves and the de Broglie equation in order to create a mathematical model to describe the distribution of electrons in an atom. This is a powerful model of the atom that was developed by Erwin Schrodinger in 1926. This model describes regions in space or orbitals as locations where electrons are most likely to occur. Schrodinger model is a model in chemistry that assumes that an electron is a wave. Bohr model fails to explain the atomic spectra of larger atoms.It could not explain the Stark effect (effect of electric field on the atomic spectrum).The Bohr model could not explain the Zeeman effect (effect of magnetic field on the atomic spectrum).Apart from that, Bohr used Plank’s constant to calculate the energy of the energy levels of the atom.īut there were a few drawbacks of the Bohr model when explaining the atomic structure of atoms other than Hydrogen.
Generally, Bohr’s model perfectly fits the hydrogen atom, which has a single electron and a small positively charged nucleus. Electrons can move from one energy level to another by absorbing or releasing energy.When an electron is moving in a certain orbital, the energy of that electron is constant.The atom is completely stable when electrons are at the lowest energy level. The smallest orbit has the lowest energy.The energy of an orbital is related to its size.Each orbit has a different radius and is named from the nucleus to the outside as n=1, 2, 3, etc., or n= K, L, M, etc., where n is the fixed energy level number.The electrons move around the nucleus in spherical orbitals, which have a fixed size and energy.Since there are discrete lines in the line spectra, Bohr stated that the orbitals of an atom have fixed energies, and electrons can jump from one energy level to the other emitting or absorbing energy, resulting in a line in the line spectra.įigure 01: Bohr Model Main Postulates of the Bohr Model This phenomenon was suggested by observations of the line spectra for the hydrogen atom. This model also states that these shells have different energies and are spherical in shape. However, the Bohr model is highly advanced than the Rutherford model since it suggests that electrons always travel in specific shells or orbits around the nucleus. Summary – Bohr vs Schrodinger Model What is Bohr Model?īohr model, which is a modification of the Rutherford model, explains the structure of the atom as composed of a nucleus surrounded by electrons.
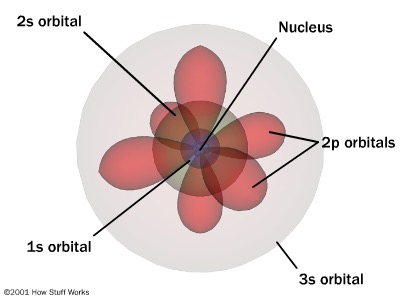
Bohr vs Schrodinger Model in Tabular Formĥ. The key difference between Bohr and Schrodinger model is that in the Bohr model, electrons are particles that occupy only certain orbits of fixed energy around the nucleus, whereas, in the Schrodinger model, electrons behave as standing waves having a greater probability of being in some areas of space compared to others.īohr model and Schrodinger model are very important models in chemistry as they describe the chemical structure of atoms and the location of electrons at a given time.
